Marine Snow: dead organisms and poop as manna in the ocean
Edit: Republished at the American Society for Microbiology blog, Small Things Considered
“When I think of the floor of the deep sea…I see always the steady, unremitting, downward drift of materials from above, flake upon flake, layer upon layer…the most stupendous “snowfall” the earth has ever seen.”
-Rachel Carson, The Sea Around Us (1951)
![]() Marine snow: does the phrase make you think of fish wearing ice skates, seahorses in knit caps, and crabs building snow-fish? If so, I can’t entirely blame you: the stuff looks like snow, hence its name. But marine snow is not composed of frozen water, each flake unique and beautiful. It is, in fact, made up entirely of dead organisms, poop, and random junk floating in the ocean. (Still fancy a marine snowball fight?)
Marine snow: does the phrase make you think of fish wearing ice skates, seahorses in knit caps, and crabs building snow-fish? If so, I can’t entirely blame you: the stuff looks like snow, hence its name. But marine snow is not composed of frozen water, each flake unique and beautiful. It is, in fact, made up entirely of dead organisms, poop, and random junk floating in the ocean. (Still fancy a marine snowball fight?)
To undergo photosynthesis, marine phytoplankton (the “wandering plants” in Greek) can only live in the sunlit areas (the photic zone) of the ocean, which rarely reaches a depth greater than 200 meters. The phytoplankton do their thing, you know, generate nearly half of the planet’s primary production (no big deal?!), until they are either eaten or die. And when you’re living at the top of a miles-deep ocean, the only way to go is down. And thus we have marine snow.
Marine snow is complicated stuff. While initially it is mostly composed of dead material, slowly drifting downward, overtime it is affected by many processes that create an indescribable mass of fluff and particles and organisms stuck together. But along the way, it continues to sink, until eventually it reaches the seafloor, often miles down. And it needs to reach that destination, as it is a primary source of carbon for deep sea organisms who have no light by which to photosynthesize.

Rattail swimming out of a cloud of marine snow near the bottom of the northern trench of the Charlie-Gibbs Fracture Zone (4400 m, June 2003) - Taken as part of MAR-ECO project through CFML
A floater or a sinker?
But how does a clump of algae or cells manage to sink for miles? By straight physics alone, a particle in still seawater would take years to reach the seafloor. Add to that waves, currents, water stratification, and you have some particles that aren’t going anywhere. Many studies have provided evidence that most movement takes place when the phytoplankton have been ingested and egested as fecal pellets (i.e. poop).
An environmentally sobering study done in 1987 and published in Nature presents strong evidence for the increased sinking rates of fecal pellets. After the Chernobyl Disaster in 1986, when a nuclear power plant blew out and released radioactive fallout, scientists studying vertical transport in the ocean decided to make the best of the tragedy. Several days after the disaster on April 26, 1986, rain fell on the ocean, delivering the radioactivity to the surface of the water. The scientists had already set up particle traps at 200 meters depth and, upon review, found that radioactivity was found in the traps just 7 days after the rainfall, with an average sinking rate of 29 m/day. Physics predicted that radioactive particles would take years to reach this depth! To explain this discrepancy, the authors identified the particles as fecal pellets and – lo and behold! – the high sinking rate was explained by the repackaging of these radioactive particles through ingestion and then egestion by zooplankton. The fecal pellets were larger in size and more dense, and thus able to sink more rapidly through the water column.
But not all fecal pellets are created equal; many factors contribute to their size and density, which in turn determines how fast they will sink and quickly they will deliver carbon and nutrients to deeper waters. Different “types” of feeders, for example, have different efficiencies of repackaging as particles. Carnivores tend to eat larger food and make it smaller, while grazers such as filter feeding mollusks or some zooplankton species collect tiny, slow-sinking particles and collect them into a larger fecal pellet, greatly increasing the sinking rate of those particular particles.
In the below figure, from a 2001 Bioscience paper entitled “Feces in Aquatic Ecosystems,” data on sinking rates of various taxa are shown. However, these data need to be taken with a grain of salt. These are sinking rates from individual studies taken in different years in different parts of the ocean. Fecal pellet sinking rate is determined not only by repackaging efficiency by species, but also by the organisms’ diets, water turbulence at the surface, water stratification, season, and even time of day. But what it does show is how variable sinking rates are, even for something as simple as a little speck of poop.

Sinking rates of fecal pellets of different taxa. Image from Wotton et al. (2001). Numbers at the end of taxa names refer to the paper data originates, which is detailed at the end of this post.
But the wild trip isn’t over for organic carbon bound up in fecal pellets. As they sink, they get stuck in larger pieces of marine snow. They start to degrade from friction alone, but also from grazing organisms or microbes that are living on them*, creating more drag and slowing their sinking rate. And then, inevitably, a fish or a zooplankter comes along and eats them, transporting the carbon back up to the surface or down deeper until they are re-released as a fresh pellet, and the cycle begins again.

Salps, a zooplankton species, can form huge schools (275 individuals per cubic meter!), and are well known for their voracious feeding on phytoplankton. They are incredibly efficient repackagers. Their fecal pellets sink rapidly, and the bodies of the dead salps themselves provide a large amount of carbon to the seafloor. (See Iseki 1981, citation at bottom)
It seems like such a long and hard way to travel: passively drifting for miles, eaten and pooped out over and over again. How does marine snow ever make it to the seafloor? We had photographs of marine snow at the seafloor, but its existence did not seem to make sense. Paper after paper reported that marine snow in the water column decreases with depth due to much grazing in the euphotic and mesopegalic (where light reaches, but not enough for photosynthesis) zones. Most papers rely on sediment traps: essentially a huge funnel to collect small particles. A 1993 Nature paper evaluated this method itself, and suggested that it severely underestimates the volume of marine snow in the water column. But there had been no detailed studies yet quantifying marine snow volume in multiple locations and depths.
That is… until this May (PNAS, 2010), when the marine snow veterans Alexander Bochdansky, Hendrik van Aken, and Gerhard Herndl decided to try a new method: quantification through photographic analysis and optical backscatter.
On a research vessel, the team traversed a 4000 km horizontal transect across the Atlantic, including area over the Romanche Fracture Zone, which at 6000 meters is the deepest section of the mid-Atlantic Ridge. The researchers threw lights at sections of water at particular angles, and the light reflected back by particles was visible in photographs (>500 um). They additionally used a Seapoint Turbidity Meter, which quantifies light reflected off of very small particles in the water through voltage. By running these tests at 17 points along their transects, and at these points, from the surface to the ocean floor, the researchers were able to visualize a bigger picture for particulate and marine snow suspension in the ocean.
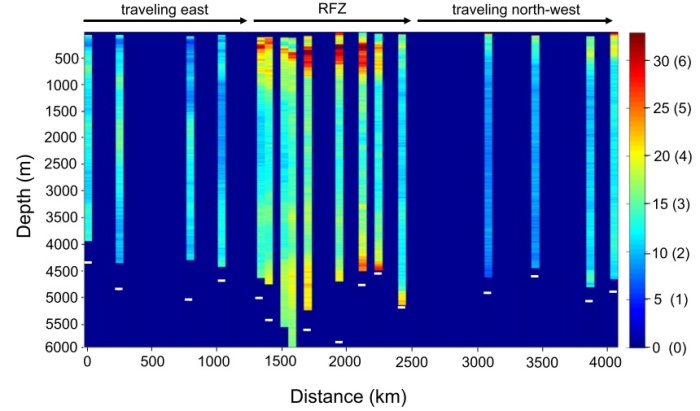
This figure shows the number of particles per frame (colored bar on right, blues indicating few pellets and warmer reds indicated high pellet number) along their 4000 km transect (x-axis) by depth (y-axis). From Bochdansky et al. 2010 (PNAS).
The figure above shows this picture. In the upper 2000 meters of the ocean, marine snow abundance is high, gradually decreasing with depth, with microscopic particles more abundant than macroscopic ones. But below 2000 meters, there is an increase in marine snow overall, and there are greater numbers of macroscopic particles than microscopic. The researchers also looked at oxygen rates in each frame they selected, and found a significant negative cross-correlation between oxygen values and particle volume, or more particles meant less oxygen. This suggests that there is greater oxygen consumption at particles, indicating microbial respiration.
The authors suggest that, at greater depths, the marine snow particles and fecal pellets form larger aggregates, which slows their sinking rate due to increased drag and decreased density. These are larger particles that are sinking very slowly or are neutrally buoyant, and are thus not caught in sediment traps. The particles that are able to escape the highly grazed areas of the mesopelagic clump together in the deep, but are less rapidly consumed because there are simply fewer organisms around to consume them. Slowly but surely they sink down, providing carbon to the depths and forming “hotspots of biological activity.”
In light of recent research: ecosystem effects of decreased phytoplankton
Marine phytoplankton have been hot in the news lately: Daniel Boyce and Boris Worm published a study in Nature (covered well by others) reporting that over the last century, marine phytoplankton biomass has decreased by 1% per year. You read right: the organisms that produce nearly half of our oxygen and absorb a significant amount of carbon dioxide are disappearing at an alarming rate.
Much of the coverage has focused on the climate change aspect of this study. Phytoplankton are a huge carbon sink, and it was assumed that increased temperatures through climate change would actually increase their growth. But – alas! – the decrease in phytoplankton biomass actually correlated with increased sea surface temperatures. So the exact opposite of what we predicted would happen is occurring, according to this study.
But I’m an ecologist: what does this decrease in phytoplankton mean for the marine ecosystem? As I’ve explained throughout this post, phytoplankton are the primary energy source for the ocean. They themselves convert sunlight to energy, are eaten by other organisms, and then this poop slowly sinks to the deep-sea to provide food for organisms living on the seafloor. In his 2002 review, Jefferson Turner argues that seasonal mass sinks of phytoplankton and algae biomass are vital for survival for deep-sea organisms.
While we probably haven’t lost enough phytoplankton to cause great loss in the deep, it’s something we should be thinking about. A tenet of ecology is that a change in the abundance of one organism can have cascading effects through an ecosystem, in turn affecting the abundances of many others. The thought of losing a primary carbon sink is unsettling; but the thought of removing a crucial food source for most of the ocean is far more horrifying. It seems unlikely enough that a fecal pellet would reach the deep on its own even with the relative abundance of phytoplankton that exists now. Losing a significant amount of phytoplankton could have grave impacts through the entire ocean ecosystem. I don’t think we want to know the end of that story.
* Fecal pellets as microhabitats or even islands is a fascinating subject. A 1978 Science paper found that there is greater taxonomic diversity on pellets than in the open water, and that the species living in and on pellets is significantly different that those in the water. Also, organisms that survive gut passage (microbes and invertebrates) are often released, live, in fecal pellets. For more on this, see this recent paper on applying island biogeography to fecal pellets.
Fecal pellet sinking rates figure:
(1) Turner JT. 1977. Sinking rates of fecal pellets from the marine copepod Potella meadii. Marine Biology 40: 249–259.
(2) Alldredge AL, Gotschalk CC, MacIntyre S. 1987. Evidence for sustained residence of macrocrustacean fecal pellets in surface waters off Southern California. Deep-Sea Research Part A: Oceanographic Research Papers 34: 1641–1652.
(3) Smayda TJ. 1969. Some measurements of the sinking rate of fecal pellets. Limnology and Oceanography 14: 621–625.
(4) Deibel D. 1990. Still-water sinking velocity of fecal material from the pelagic tunicate Dolioletta gegenbauri. Marine Ecology Progress Series 62: 55–60.
(5) Ladle M, Welton JS, Bell MC.1987. Sinking rates and physical properties of faecal pellets of freshwater invertebrates of the genera Simulium and Gammarus. Archiv für Hydrobiologie 108: 411–424.
(6) Fowler SW, Small LF. 1972.Sinking rates of euphausiid fecal pellets. Limnology and Oceanography 17: 293–296.
(7) Ladle M, Welton JS, Bell MC.1987. Sinking rates and physical properties of faecal pellets of freshwater invertebrates of the genera Simulium and Gammarus. Archiv für Hydrobiologie 108: 411–424.
(8) Yoon WD, Marty JC, Sylvain D, Nival P. 1996.Degradation of faecal pellets in Pegea confoederata (Salpidae,Thaliacea) and its implications in the vertical flux of organic matter. Journal of Experimental Marine Biology and Ecology 203: 147–177.
(9) Taghon GL, Nowell ARM, Jumars PA. 1984. Transport and breakdown of fe- cal pellets: Biological and sedimentological consequences. Limnology and Oceanography 29: 64–72.
Bochdansky, A., van Aken, H., & Herndl, G. (2010). Role of macroscopic particles in deep-sea oxygen consumption Proceedings of the National Academy of Sciences, 107 (18), 8287-8291 DOI: 10.1073/pnas.0913744107
Boyce, D., Lewis, M., & Worm, B. (2010). Global phytoplankton decline over the past century Nature, 466 (7306), 591-596 DOI: 10.1038/nature09268
Fowler, S., Buat-Menard, P., Yokoyama, Y., Ballestra, S., Holm, E., & Nguyen, H. (1987). Rapid removal of Chernobyl fallout from Mediterranean surface waters by biological activity Nature, 329 (6134), 56-58 DOI: 10.1038/329056a0
Goldthwait, S., Carlson, C., Henderson, G., & Alldredge, A. (2005). Effects of physical fragmentation on remineralization of marine snow Marine Ecology Progress Series, 305, 59-65 DOI: 10.3354/meps305059
Iseki, K. (1981). Particulate Organic Matter Transport to the Deep Sea by Salp Fecal Pellets Marine Ecology Progress Series, 5, 55-60 DOI: 10.3354/meps005055
Lampitt, R., Hillier, W., & Challenor, P. (1993). Seasonal and diel variation in the open ocean concentration of marine snow aggregates Nature, 362 (6422), 737-739 DOI: 10.1038/362737a0
SILVER, M., SHANKS, A., & TRENT, J. (1978). Marine Snow: Microplankton Habitat and Source of Small-Scale Patchiness in Pelagic Populations Science, 201 (4353), 371-373 DOI: 10.1126/science.201.4353.371
Turner, J. (2002). Zooplankton fecal pellets, marine snow and sinking phytoplankton blooms Aquatic Microbial Ecology, 27, 57-102 DOI: 10.3354/ame027057
WOTTON, R., & MALMQVIST, B. (2001). Feces in Aquatic Ecosystems BioScience, 51 (7) DOI: 10.1641/0006-3568(2001)051[0537:FIAE]2.0.CO;2






[…] beautiful marine snowfall you see in deep-sea videos is really kinda gross, and yet it is a essential to ocean health and carbon […]
ResearchBlogging.org News » Blog Archive » Editor’s Selection: Wright Stuff, Snow?, Octopusomics, and Gran’s Influence
August 19, 2010 at 4:05 pm
[…] And sometimes it is poop of one animal feeding another, as Hannah Waters, of Culturing Science, discovered: Marine Snow: dead organisms and poop as manna in the ocean. […]
Carnal Carnival #1 – Essentials of Elimination | A Blog Around The Clock
August 20, 2010 at 7:04 am
[…] Culturing Science: Octopuses doing tricks on the internet and our search for non-human ‘intelligence’ Culturing Science: Why Scientists Should Read Science Fiction Culturing Science: Microbe biogeography: the distribution, dispersal and evolution of the littlest organisms Culturing Science: Marine Snow: dead organisms and poop as manna in the ocean […]
Open Laboratory 2010 – submissions so far | A Blog Around The Clock
August 30, 2010 at 8:12 am
[…] Waters from Culturing Science gives us Marine Snow: dead organisms and poop as manna in the ocean. It is a well-known fact that any blog post with poop in the title is likely to be good […]
killer shrimp : : Carnival of the Blue XL - Hit Stuff!
September 15, 2010 at 12:02 pm
[…] conditions are right, will form huge blooms which eventually will sink rapidly to the seafloor as marine snow, providing carbon to the deep sea. Coccolithophores have calcium carbonate shells and thus affect […]
The grand diversity of marine phytoplankton species: focusing from space « Sleeping with the Fishes
October 27, 2010 at 2:05 pm
[…] Culturing Science: Octopuses doing tricks on the internet and our search for non-human ‘intelligence’ Culturing Science: Why Scientists Should Read Science Fiction Culturing Science: Microbe biogeography: the distribution, dispersal and evolution of the littlest organisms Culturing Science: Marine Snow: dead organisms and poop as manna in the ocean […]
Open Laboratory 2010 – one month left to submit! | A Blog Around The Clock
November 1, 2010 at 8:11 am
[…] the distribution, dispersal and evolution of the littlest organisms Culturing Science: Marine Snow: dead organisms and poop as manna in the ocean Culturing Science: Inevitability and Oil, Pt. 1: the inherent risk for accidents in complex […]
Open Laboratory 2011 – three weeks to go! | A Blog Around The Clock
November 8, 2010 at 1:53 pm
[…] the distribution, dispersal and evolution of the littlest organisms Culturing Science: Marine Snow: dead organisms and poop as manna in the ocean Culturing Science: Inevitability and Oil, Pt. 1: the inherent risk for accidents in complex […]
Open Laboratory 2010 – two weeks to go! | A Blog Around The Clock
November 15, 2010 at 9:14 am
[…] the distribution, dispersal and evolution of the littlest organisms Culturing Science: Marine Snow: dead organisms and poop as manna in the ocean Culturing Science: Inevitability and Oil, Pt. 1: the inherent risk for accidents in complex […]
Open Laboratory 2010 – only eight days till the deadline! | A Blog Around The Clock
November 22, 2010 at 3:46 pm
[…] the distribution, dispersal and evolution of the littlest organisms Culturing Science: Marine Snow: dead organisms and poop as manna in the ocean Culturing Science: Inevitability and Oil, Pt. 1: the inherent risk for accidents in complex […]
Only three days to go – Open Laboratory final stretch for submissions! | A Blog Around The Clock
November 27, 2010 at 11:20 am
[…] the distribution, dispersal and evolution of the littlest organisms Culturing Science: Marine Snow: dead organisms and poop as manna in the ocean Culturing Science: Inevitability and Oil, Pt. 1: the inherent risk for accidents in complex […]
It’s getting hot – submissions for Open Laboratory 2011 are flying in by the dozens per hour… how about you? | A Blog Around The Clock
November 28, 2010 at 10:55 am
[…] the distribution, dispersal and evolution of the littlest organisms Culturing Science: Marine Snow: dead organisms and poop as manna in the ocean Culturing Science: Inevitability and Oil, Pt. 1: the inherent risk for accidents in complex […]
Open Laboratory 2010 – the final stretch! | A Blog Around The Clock
November 29, 2010 at 9:03 am
[…] the distribution, dispersal and evolution of the littlest organisms Culturing Science: Marine Snow: dead organisms and poop as manna in the ocean Culturing Science: Inevitability and Oil, Pt. 1: the inherent risk for accidents in complex […]
Open Laboratory 2010 – submissions now closed – see all the entries | A Blog Around The Clock
December 1, 2010 at 3:08 am
[…] I regret to inform you that tonight’s reading has been canceled due to weather and the inability of planes to get me from Denver to New York. We look forward to re-scheduling in the near future, and in the meantime encourage you to step outside, look up at the snow, and imagine that each white flake is a piece of poop. Because if you were in the ocean, that’s what that falling, snow-like morass would be. (More here: https://culturingscience.wordpress.com/2010/08/19/marine-snow/) […]
» Jonathan Swift Poo-etry Event CANCELED The Poop Project
December 27, 2010 at 1:40 pm
[…] Organic matter from surface waters falls into the deep sea; there is the possibility that these fluxes of sustenance may show lunar patterns. Previous work has shown growth bands in some species of deep-sea corals that may correspond to monthly or lunar periods. Other hypotheses include the idea that these animals can somehow directly perceive moonlight at great depths, or that deep tidal (related to lunar phase) currents exist. […]
Lunar cycles and reproduction in the deep sea « Uncharted Atolls
February 26, 2011 at 9:23 pm
[…] Marine snow: dead organisms and poop as manna in the ocean […]
I have not abandoned you. « Culturing Science – biology as relevant to us earthly beings
May 25, 2011 at 3:39 pm
[…] Don’t go eating that yellow snow: The organic particulates that rain down from the productive surface waters into the oceans deeps have been, rather poetically, referred to as ‘marine snow’. For the full health warning read Hannah Waters in Culturing Science, August 19, 2010 […]
Lancashire MCS Blog » Blog Archive » Marine science update 19th June 2011
June 19, 2011 at 1:45 pm
I’m lucky enough to live on the West Coast of South Australia which is lapped by the pristine waters of the Southern Ocean.
As a keen fisherman, this is paradise. But the concept of marine snow, and its significance to the ocean, is a new one for a non-scientist like myself.
I’m conditioned to seeing the good get better as it moves from the bottom to the top e.g. “the cream always rises to the top,” I’ve been taught.
If I fully comprehended your post, in the sea the good stuff, whether plankton remains, or bird poop, ends up as vital fodder to fuel the world below.
Thanks for the illuminating article. I’ll look at the water surface with new eyes next time I’m out there waiting for the fish to bite.
Thankfully there are no concerns of radioactive fallout in this part of the world, and the marine snow remains pure.
Leon
January 7, 2014 at 9:44 pm